|
| Friday, 16 August 2019, 21:19 HKT/SGT | |
| |
|
|
|
|
-Turnover (excl. the effect of "two-invoice system") up 14.4% to RMB 3,401.5 Million
-Profit up 22.2% to RMB 1,167.5 Million
-Focused on Innovative Research and Development
-Built Product Clusters of Innovative Patented Products and Complex Generic Drugs
-Expanded the Core Competitive Advantages
-Deepened Academic Network of Existing Products & Expanded the Retail Coverage
-Achieved a Sound Growth |
HONG KONG, Aug 16, 2019 - (ACN Newswire) - China Medical System Holdings Limited ("CMS", or the "Company", SEHK:867), together with its subsidiaries (the "Group"), a well-established, innovation-driven specialty pharma with a focus on sales and marketing in China, announced its unaudited interim results for the 6 months ended 30 June 2019 ( the "Reporting Period").
 |
 | | *European Medicines Agency ("EMA") * * Therapeutic Goods Administration ("TGA") |
Performance Highlights:
- Maintained the Steady Growth: Turnover up 11.7% to RMB2,964.4 million; if excluding the effect of the "two-invoice system", turnover up 14.4% to RMB3,401.5 million. Profit for the period up 22.2% to RMB1,167.5 million
- Existing Products Achieved Continuous Growth: By adhering to the academic-oriented differentiation promotion and deeply exploring products' evidences of evidence-based medicine, each product-line achieved good growth
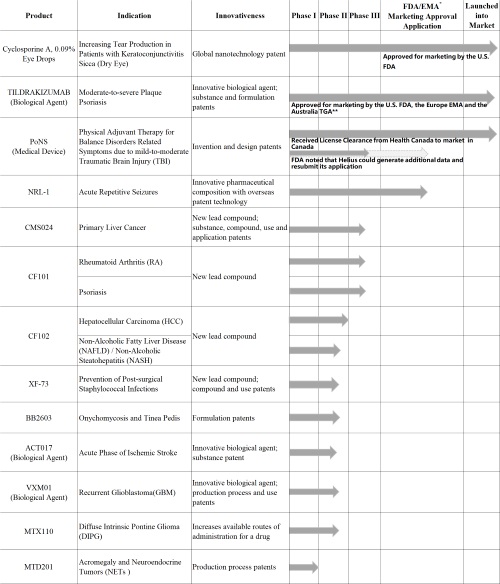
- Accelerated Innovative R&D: During the Reporting Period, the Group acquired four innovative patented products. Among them, two have been launched in the U.S., the E.U. and other regions, and the other two are at the clinical research stages, which expanded the number of the Groups' innovative products to thirteen
- Strategically Planned and Upgraded Existing Promotion Network: Optimized organizational structure of academic network and steadily promoted the coverage of retail network; for the six months ended 30 June 2019, the Group's promotion network had covered over 57,000 hospitals and medical institutions in China
During the Reporting Period, by continuously optimizing academic network, the Group further enhanced its products' academic influence, while utilizing its high-efficient and compliant operation management system, the Group achieved a sound growth. Turnover was up 11.7% to RMB2,964.4 million (H1 2018: RMB2,655.0 million); excluding the effect of the "two-invoice system", turnover up 14.4% to RMB3,401.5 million (H1 2018: RMB2,974.3 million). Profit for the period recorded an increase of 22.2% to RMB1,167.5 million (H1 2018: RMB955.1 million). Declared interim dividend was up 22.6% to RMB0.1883 per share (H1 2018: RMB0.1536).
Mr. Lam Kong, Chairman and Chief Executive of CMS, said: "The soul of a pharmaceutical company for its survival and development is its products. A product portfolio with differentiated competitive advantages is the most important strength for the Group to become a global innovative specialty pharmaceutical company. Facing the reshaping period of the pharmaceutical industry, the Group has never forgotten its original aspiration. On the one hand, with an international vision, the Group has actively made arrangement of innovative products that can provide new treatment options for Chinese patients, in order to consolidate and enhance its product advantages and competitiveness. On the other hand, the Group deepened national academic network to solidify the academic-oriented differentiation of its existing products while further expanding their academic influence, which helped the Group to achieve a sound growth during the Reporting Period."
Driving Force for the Development
With the innovative research and development as its core strategy, the Group has concentrated on the arrangement of innovative patented products. At the same time, the Group has actively made arrangement of complex generic drugs with high imitation barriers, and carried out the strategic collaboration with leading generic drug company on complex generic drugs. Both the innovative patented products and complex generic drugs are expected to enhance the key competitive advantage of the Group. In addition, the Group carried out the strategic arrangement of generic drugs with sufficient market competitiveness. By taking advantages of advanced pharmaceutical techniques, high quality standards and guaranteed supply capabilities of overseas mature pharmaceutical companies, the Group expected to directly introduce the overseas-launched generic drugs portfolio with proven quality and affordable price to domestic market under a relatively light-asset model. The Group is going to build the sustainable development driving force with the comprehensive arrangement of innovative patented products, complex generic drugs clusters with high imitation barriers and generic drugs clusters with sufficient market competitiveness.
During the Reporting Period, the Group in-licensed two overseas-launched innovative patented products from Sun Pharmaceutical Industries Ltd., India's top pharmaceutical company, which are the globally first 0.09% cyclosporine ophthalmic eye drops using nanotechnology for the treatment of dry eye, and a monoclonal antibody that specifically targets interleukin-23 for the treatment of plaque psoriasis. The Group also made an equity investment in Midatech Pharma PLC, a U.K. specialty pharmaceutical company focused on R&D of a pipeline of medicines for oncology and immunotherapy, and obtained its two innovative products and three innovative technology platforms.
As at 30 June 2019, the Group owned thirteen innovative products in various fields including ophthalmology, dermatology, nervous system, anti-tumor, immune system, digestive system, anti-infection and endocrine system. The development process of innovative products is listed as Photo2.
Existing Products Development
The Group's existing products mainly include products under cardio-cerebrovascular line (mainly include XinHuoSu, Plendil and Deanxit), digestion line (mainly include Ursofalk, Salofalk, Bioflor and Combizym), ophthalmology line (mainly include Augentropfen Stulln Mono Eye Drops) and dermatology line (mainly include Hirudoid). Through the line-divided academic-oriented promotion of its existing products, the Group deeply explored products' market potentials while enhancing the synergistic effect of products and working efficiency of its promotional staff. During the Reporting Period, the Group's product lines achieved good growth. Among them, compared with the same period last year, the revenue of products under cardio-cerebrovascular line would increase by 14.7% to RMB1,940.4 million if excluding the effect of the "two-invoice system"; the revenue of products under digestion line increased by 19.6% to RMB1,027.2 million; the revenue of products under ophthalmology line increased 9.5% to RMB115.2 million; the revenue of products under dermatology line increased by 16.5% to RMB86.0 million.
Network Development
During the Reporting Period, the Group optimized the organizational structure of promotion network, making its product promotion more refined and professional. At the same time, with the full use of digital tools and digital management system, the Group achieved a new academic promotion model with integration of on-line and off-line to make the operational data more analyzable and traceable. Meanwhile the management of employee behavior was more standardized, ensuring the compliance of the Group's academic promotion. The Group improved the execution and efficiency of employees, aiming to build a professional, dedicated and quality promotional team so as to accumulate energy for the promotion of its upcoming innovative products in China. For the six months ended 30 June 2019, the Group's promotion network had covered over 57,000 hospitals and medical institutions in China.
At the same time, the Group steadily promoted the construction and development of its retail team, maintained and expanded the retail coverage and channel layout. Through improving the compensation system, completing the standards and processes of the retail business, and optimizing the retail data management system, the Group achieved an enhanced internal management system of retail business to lay the foundation for the expansion and development of its retail team.
Outlook and Future Development
China's pharmaceutical market is huge and expanding rapidly driven by factors such as population ageing, urbanization, wealth-increasing and comprehensive coverage of various medical insurance policies. The Group believes that China's pharmaceutical market is endowed with promising growth potential in the future. With the ever-growing professional academic promotion capability and the sustainable revenue contribution of its existing products, the Group has confidence in promoting the long-term strategic arrangement of diversified innovative products with different developmental stages and mixed risk levels, in order to maintain a steady growth of its performance.
Mr. Lam Kong concluded, "Looking ahead, innovation in China's pharmaceutical market will see its full boom in the near future. The Group will continue to constantly arrange and develop innovative patented products globally while seizing the opportunities to make arrangement of complex generic drugs, forming these products with highly competitive barriers into products lines and gradually products clusters, in order to enhance the Group's innovative competitiveness in China's pharmaceutical industry. Meanwhile, the development of the generic drugs clusters with sufficient market competitiveness will ensure the product portfolio's dynamic supplement and iteration. Both the Group's generics clusters and the existing products will support its innovative products' long-term strategic deployment with strong economic contribution. In the meantime, the Group will continuously upgrade its academic network platform to provide a carrier for the development of its upcoming innovative products in China, maintaining the Group's sustainable growth. The Group dedicates to becoming a leading innovation-driven specialty pharmaceutical company and believes that success belongs to the brave. Pharmaceutical companies which embrace changes courageously and take actions immediately to create opportunities will be able to promote their development while keeping forging ahead and moving forward in the pharmaceutical industry! "
About China Medical System Holdings Limited
China Medical System Holdings Limited is a well-established, innovation-driven specialty pharma with a focus on sales and marketing in China. Through global strategic collaboration, CMS accelerates the strategic arrangement focusing on innovative products, for establishing product clusters with highly competitive barriers, to contribute the long-term sustained driving force for the Group's development.
Topic: Press release summary
Sectors: Daily Finance, Daily News, Healthcare & Pharm
https://www.acnnewswire.com
From the Asia Corporate News Network
Copyright © 2024 ACN Newswire. All rights reserved. A division of Asia Corporate News Network.
|
|
|
|

|
|
|
| |
Latest Press Releases
TIME Interconnect Technology Limited Announces Final Results For The Nine Months Ended 31 December 2023
Apr 26, 2024 18:26 HKT/SGT
|

|
|
UK advertising reports GBP36.6bn spend in 2023 
Apr 26, 2024 17:30 HKT/SGT
|

|
|
CanSinoBIO CSO Shares the Latest Results of the Company's Globally Innovative Pneumococcal Vaccine
Apr 26, 2024 15:36 HKT/SGT
|

|
|
Edvantage Group Announces FY2024 Interim Results 
Apr 26, 2024 13:43 HKT/SGT
|
|
|
Internationally Renowned Botulinum Toxin Experts Join WizMedi Bio's New Botulinum Toxin Development Project 
Apr 26, 2024 13:27 HKT/SGT
|

|
|
Imexpharm Corporation Hosts Shareholders, Analysts and Potential Investors at its 2024 Annual General Meeting
Apr 26, 2024 13:16 HKT/SGT
|

|
|
JCB enables JCB Contactless acceptance at Taichung MRT in Taiwan 
Friday, April 26, 2024 9:00:00 AM
|

|
|
Jeff Martin Auctioneers to Manage Sale of Sabine Mining Company Assets for North American Coal
Apr 26, 2024 08:00 HKT/SGT
|

|
|
Cambridge Isotope Laboratories (CIL) Shows Long-Term Commitment to Xenia, Ohio, Facility With New Land Purchase
Apr 25, 2024 21:00 HKT/SGT
|

|
|
Black Spade Donates Art and Jewellery Collection To Hong Kong Red Cross 
Apr 25, 2024 20:40 HKT/SGT
|

|
|
|
|
More Press release >> |
|
|
|